
The exact biological role of caffeine and related purine alkaloids of plants is still unclear, although there are now two main hypotheses. The pharmacological actions are thought to be mediated via blockade of the adenosine A 1 and A 2A receptors ( Cauli and Morelli, 2005). These key residues are probably functionally important and will guide future studies with implications for the biosynthesis of caffeine and its derivatives in plants.Ĭaffeine is a plant alkaloid known to have sensory and stimulatory effects when consumed in beverages such as coffee ( Coffea arabica and Coffea canephora) and tea ( Camellia sinensis). A phenylalanine-266 to isoleucine-266 change in DXMT is also likely to be crucial for the discrimination between mono and dimethyl transferases in coffee. Likewise, a change from glutamine-161 in XMT to histidine-160 in DXMT is likely to have catalytic consequences. 
Serine-316 in XMT appears central to the recognition of XR. Our structures reveal several elements that appear critical for substrate selectivity. Both were cocrystallized with the demethylated cofactor, S-adenosyl- l-cysteine, and substrate, either xanthosine or theobromine. canephora ‘robusta’, xanthosine (XR) methyltransferase (XMT), and 1,7-dimethylxanthine methyltransferase (DXMT). 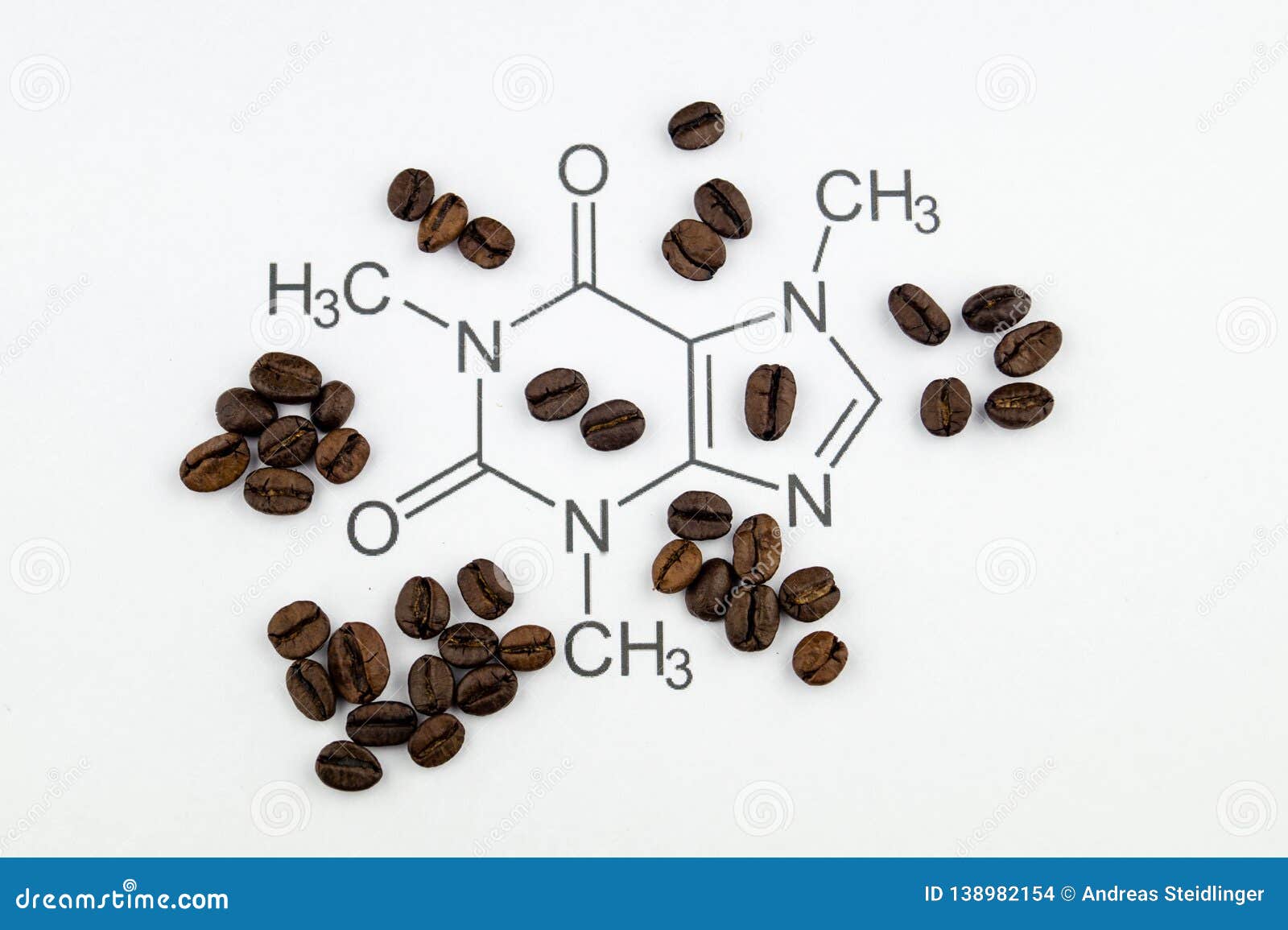
Here we describe the structures of two S-adenosyl- l-methionine-dependent N-methyltransferases that mediate caffeine biosynthesis in C. Caffeine (1,3,7-trimethylxanthine) is a secondary metabolite produced by certain plant species and an important component of coffee ( Coffea arabica and Coffea canephora) and tea ( Camellia sinensis).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
